
Laser Safety for Clinical Applications
Laser Hazards
Higher power therapeutic lasers for neuromusculoskeletal therapy are becoming more common as a primary therapy device rather than an adjunctive treatment. Although high power lasers have a significant advantage for penetration in deep musculoskeletal tissues and shorter treatment times, the potential risk for injury is greater with high power lasers because of the greater energy density per unit area as compared to low power class III lasers. Class IIIb lasers are those ranging from 5-500mw. Class IV lasers have power ratings over 500mw in continuous wave or energy density of 10 J/cm2 in pulsed modulation. (1) In musculoskeletal therapy, however, the class IV laser power ranges from 500mw to 12,000mw. Regardless of power, providers must pay attention to laser safety.
As with all therapeutic procedures, some element of risk is present through negligence or accident. For example, additional musculoskeletal injury or exacerbation may occur from premature or excessive application of therapeutic exercise. Burns from hot packs and galvanic stimulation are known to occur. Deep tissue burns and tissue cavitation are possible from ultrasound and diathermy. Frostbite occurs from cryotherapy and excessive spray and stretch technique. Infection hazards are often overlooked from bacterial and fungal cross-contamination occurring with poorly sanitized equipment. These hazards are easily prevented or reduced with safety protocols for each application. Laser therapy is extremely safe when basic protocols are followed by the laser operator and support staff.
The primary reason for following laser safety measures is the potential for thermal injury to the retina with direct or reflected laser. The lens of the eye effectively magnifies laser beam thus, a relatively low wattage of laser has the potential of damaging the retina with brief exposure. A laser beam of sufficient power can produce retinal damage when the beam enters the lens and is magnified on the retina surface. With higher power lasers, permanent blindness can be the result.
The blink reflex protects the eye when a visible light flash occurs however when the light source is an invisible high watt infrared source, the protective blink reflex is not elicited. Here in lies the primary caution with most therapeutic diode lasers; the laser beam generated is within the invisible infra-red spectrum. Thermal injury to the superficial skin is also a hazard with higher wattage lasers such as the class IV infrared therapy lasers, however, thermal skin injury is more common with surgical-type lasers.
Thermal damage may occur when a high degree of beam collimation is used. Modifying a laser tip to reduce the size of the beam also increases the energy density of the beam. Significant photonic energy is present with a class IV laser and a small spot size. Thermal injury to the skin may occur if contact with cutaneous tissues carelessly exceeds normal treatment parameters. A fire hazard may also be present with class IV lasers due to the high heat and fluence of the beam. However, when obvious sources of potential ignition are removed from the laser treatment area such as open volatile chemicals, fire hazards are only possible from highly unusual equipment failure or extremely careless laser usage. Never direct a class IV laser at, or toward dark surfaces, paper or plastic; they may catch fire or be damaged from the high temperatures generated by the laser beam.
Laser Safety Officer
Every clinic using a laser should have an individual trained in the safe operation of laser therapy and a daily safety checklist should be observed. This individual (usually the doctor), is the Laser Safety Officer (LSO).
The laser safety officer is the individual designated in a laser facility who has the authority to monitor and enforce measures that prevent laser injury. The LSO in a laser therapy center is usually the physician or medical director who is responsible for controlling access to the laser and prevention of injury from laser exposure. The LSO restricts all non-authorized use of the laser and performs the routine safety assessments of the facility and equipment. [Table 1] The LSO inspects all goggles for cracks or deterioration and inspects the integrity of the laser device for defects that may lead to safety component failure (housing, locks, optical fiber and probe).
The laser safety officer also provides oversight of the laser operator when laser use is delegated to another health care professional, therapist or registered chiropractic assistant. The LSO also must post laser warning signs outside of the treatment area. The LSO is responsible for managing and documenting all laser-related injuries as well as documenting any laser repair history.
Precautions
Lasers should be operated in a closed room or curtained area with controlled access. Extra goggles should be kept outside the room should entry be necessary by authorized personnel during laser treatment. Glass, windows or shiny jewelry may reflect the incident beam and coverings for windows may be necessary.
Many states require registration of class IIIb and class IV lasers. It’s suggested you contact the appropriate state regulatory department, (Department of Nuclear Safety Division of Electronic Products, IDNS). In Illinois, laser registration is required. The registration form is titled Laser Registration it is free to register and the form is found online for download at https://www2.illinois.gov/iema/NRS/RadSafety/documents/laser.pdf The form is easy to complete and should be mailed to the IDNS after the purchase of any class IIIb or class IV laser.
Table 1: Laser Safety Checklist
Laser safety checklist for the laser operator and Laser Safety Officer
❑ Appropriate warning signs posted
❑ Access to the laser and treatment area is secure and controlled
❑ Visually inspect and clean all optical connectors for dirt, debris, etc.
❑ Inspect laser for proper function
❑ Visually inspect and clean all safety goggles
❑ Goggles available for all persons in Nominal Hazard Zone
❑ Extra goggles placed outside the treatment room if necessary
❑ Sources of potential laser beam reflection and scatter controlled
❑ Treatment protocol established for the patient
❑ Laser injury management protocol in place for accidental injury
❑ Document laser treatment and post-treatment outcome
Laser exposure and protective eyewear
The three most important points in laser safety are maximum permissible exposure (MPE), nominal hazard zone (NHZ), and the optical density (OD) of protective eyewear. These values must be known by the laser safety officer and laser operator. Laser manufacturers usually provide this information in the specification page of the laser operation manual.
The MPE is the level of laser radiation to which a person may be exposed without hazardous effects or adverse biological changes in the eye or skin. This is a time-based calculation versus wattage level. There is a ¼-second, 10-second, 600-second, and 30,000-second limits for manufactured devices used in industrial settings. There are 28,800 seconds in an 8-hour day. This is rounded up to 30,000 seconds. The NHZ describes a space or distance in which the level of direct, reflected, or scattered radiation during normal operation of the laser exceeds the maximum permissible exposure. The area outside the NHZ is the distance from the laser source that does not exceed the MPE. Some high powered lasers may have a nominal hazard zone exceeding 20 to 30 meters. Specular reflections from class IV lasers are hazardous if they directly enter the eye. [Figure 1]
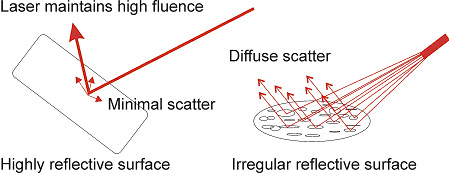
Figure 1 A) Specular and B) Diffuse reflections
Optical density (OD) is a logarithmic calculation for optical attenuation of laser provided by protective eyewear. The OD value for various lasers is computed based on the amount of attenuation needed and exposure time with the specific laser wavelength and wattage. Knowing the appropriate exposure time and distance from direct or scatter radiation, along with wattage and wavelength considerations, allow the laser safety officer to select the appropriate protective eyewear. Treatment applications with laser also determine the shape of the lens and the need for extra protection. For example, patients with smaller faces may require smaller goggles, or using the laser for sinus drainage application requires a tighter fitting goggle that seals tight against the eye socket. All personnel and patients in the nominal hazard zone must wear protective eyewear with the operation of class IIIb and IV lasers. (1)
Laser Therapy Contraindications
Contraindications to laser therapy may be absolute, relative or anecdotal/ unproven. Precautions to use are also important to consider where patients may develop side effects from laser therapy because of specific disorders or conditions.
High power laser is harmful to eyes and protective eyewear is essential for the operator and all persons present in the room. In a study of all reported laser injuries (including all laser uses), there were 339 laser accidents between the years 1984-1996. (2)With reference to surgical and industrial lasers, the vast majority of all eye injuries reported have resulted from failure to wear protective eyewear. A recent study of laser eye injury within facilities using lasers for the U.S. Department of Energy reported seven cases of eye injury since 2001; all of which resulted from lack of personal protective eyewear. (3)Although thermal damage is possible to the superficial skin when excessive laser wattage is used or from prolonged skin contact, the injury is self-limited. Biostimulative hazards are possible with laser exposure to the thyroid and various cancers. There are many theoretical biostimulative as well as over-stimulation hazards to tissues of the body from laser exposure that has not been scientifically confirmed or recognized.
Absolute contraindications to therapeutic laser are those exposures where known tissue damage may occur. [Table 2] Relative contraindications to laser use are based on specific patient status and conditions and should be decided on a case by case application. [Table 3] There are generalized precautions and unproven or anecdotal precautions. The absolute contraindications are as follows: Direct eye exposure is damaging to the retina and cornea. Permanent blindness may result. Laser should not be used directly over cancer or tumor.
The laser should not be used over the thyroid gland or over a gravid uterus. However, the use of laser away from the abdomen and uterus in the pregnant mother is safe. Relative contraindications include laser hypersensitivity and photosensitizing medications or skin creams, although there are many that react with the UV spectrum, very few react with infrared. Some autoimmune disorders and severe Lupus may cause a hypersensitivity to laser therapy.
Holding a laser over a tattoo will cause a rapid thermal effect, because of the absorption of laser on dark colors. Laser therapy in immuno-suppressed patients particularly AIDS may produce an adverse response. Patients with renal failure on dialysis may suffer side effects of nausea resulting from vasodilatation and the release of toxins into the bloodstream that cannot be eliminated. Patients with severe collagen vascular diseases should be treated in a small region for a test response, before initiating a whole body scanning technique. Laser therapy in encephalopathy; meningitis; or neuronopathy may have the potential for exacerbation when the organism or pathological process is unknown.
Table 2: Absolute Contraindications in Laser Therapy
- Eye exposure
- Over cancer or tumor
- Over thyroid
- Over gravid uterus
Table 3: Relative Contraindications in Laser Therapy
- Over tattoos
- Patients using IR photosensitizing medications
- Patients using steroids
- Autoimmune disorders
- Renal failure (severe)
- Lupus (severe)
- Encephalopathy
- Meningitis
- Neuronopathy
- Hypersensitivity to laser
Treatment/precautions include hairline sensitivity; using laser through dark clothes because of thermal heating of dark fabrics; patients on anticoagulant therapy, because of the greater potential for bruising; iodine treated wounds, because of the potential for increased absorption of laser by Betadine™; steroid therapy, and dark skin (high power red laser only). [Table 4]
Unproven contraindications for laser therapy have anecdotal evidence or an assumption of harm because of inappropriate association with another known hazard associated with a different physical therapy device. Unless the anecdotal contraindication has been disproved, some level of caution or avoidance is nevertheless suggested when using laser therapy. Unproven contraindications include using laser over ununited fontenelles and epiphyseal growth plates because of the potential for asymmetric closure.
The contraindication associated with laser and systemic infections is the potential for biostimulative growth of the organism. According to Navratil et al. (4), some of the contraindications to laser therapy that have been misreported are as follows: fever and infectious diseases; certain blood diseases; heavy blood losses; neuropathies; and irradiation in the region of gonads. Laser use in multiple sclerosis, has an anecdotal/unproven contraindication because of the potential for exacerbation from heat and the biostimulative effect on the immune system.
The contraindication of laser with epileptics is an assumed risk because of the association of photic flicker producing seizures in photosensitive individuals. Although this is known to occur with 15-65 hz flicker and pattern-based visual stimulation in susceptible individuals, it has not been shown to occur with continuous wave or high frequency modulated laser. Patients with highly unstable epilepsy should not receive laser therapy because of the risk of seizure. It is doubtful that a photosensitive stimulation risk to mild epileptic patients using high-frequency modulation or continuous wave laser would cause a seizure. The use of a class IV laser directly over a pacemaker is contraindicated; however, the surrounding use of laser in patients with a pacemaker is appropriate and demonstrated to be without risk. The contraindication to laser therapy in patients with pacemakers only remains in the literature, because of an inappropriate assumption of risk similar to ultrasound and diathermy.[Table 5]
Table 4: Precautions in Laser Therapy
- Hairline sensitivity
- Patients using anticoagulants
- Through dark clothing
- Iodine treated wounds
- Steroid therapy
- Dark skin (High power red laser)
Table 5: Unproven Contraindications in Laser Therapy
- Ununited fontanelles
- Ununited epiphyseal plate
- Systemic infections
- Multiple sclerosis
- Epilepsy (mild)
- Patients with Pacemaker
Adverse reactions to laser therapy
Mild adverse reactions with normal treatment protocols may occur.Rarely, some patients have reported increased pain; this may be more common in patients after steroid injection. Occasionally, patients may describe a bruised sensation in muscles the following day. Mild bruising also may occur from pressure of the laser wand into cutaneous tissues using the laser tip in conjunction with deep tissue myofascial techniques. Mild temporary dizziness may rarely occur, most likely from the peripheral vasodilatory effect and a possible drop in blood pressure.
Summary
Safe operation of higher power class IIIb and IV lasers is not difficult when the laser operator has basic training in laser safety. The laser operator and Laser Safety Officer should follow a laser safety check list each day before treating patients as a simple method to avoid equipment failure as a source of laser injury. Wavelength specific protective goggles are mandatory for all individuals in the nominal hazard zone or treatment room. An adequate history should be taken from the patient to determine if any contraindications to treatment are present. Following preset timed protocols for laser dosage will prevent some if not all of the potential adverse responses to treatment.
References
- OSHA Technical Manual. Section III, Chapter 6. Laser Hazards. Subsections I-VII. U.S. Department of Labor 2001.
- Brahmavar S, Hetzel F. Medical Lasers, Quality Control, Safety Standards, and Regulations. AAPM Report No. 73. AAPM General Medical Physics Committee. 2001.
- Special Operations Report: Laser Safety. U.S. Department of Energy, Office of Corporate Performance Assessment. February 2005.
- Navratil, L. Kymplova, J. Contraindications in Noninvasive Laser Therapy: Truth and Fiction. Journal of Clinical Laser Medicine & Surgery. Vol. 20, No. 6: 341-343. Dec 2002.
Jerome M. True, D.C., DABCN is a board-certified chiropractic neurologist practicing in Stuart, FL. He has extensive experience with laser therapy and neuromusculoskeletal disorders. Dr. True is the co-author of the textbook: Myelopathy, Radiculopathy and Peripheral Entrapment Syndromes, CRC Press: 2002. He has recently completed a chapter titled: “Nerve Injuries in Competitive Sports”, for the second edition of Conservative Management of Sports Injuries, Jones and Bartlett: 2007. He has also contributed to and/or illustrated many published articles on Chiropractic Neurology.



















